ABCNews
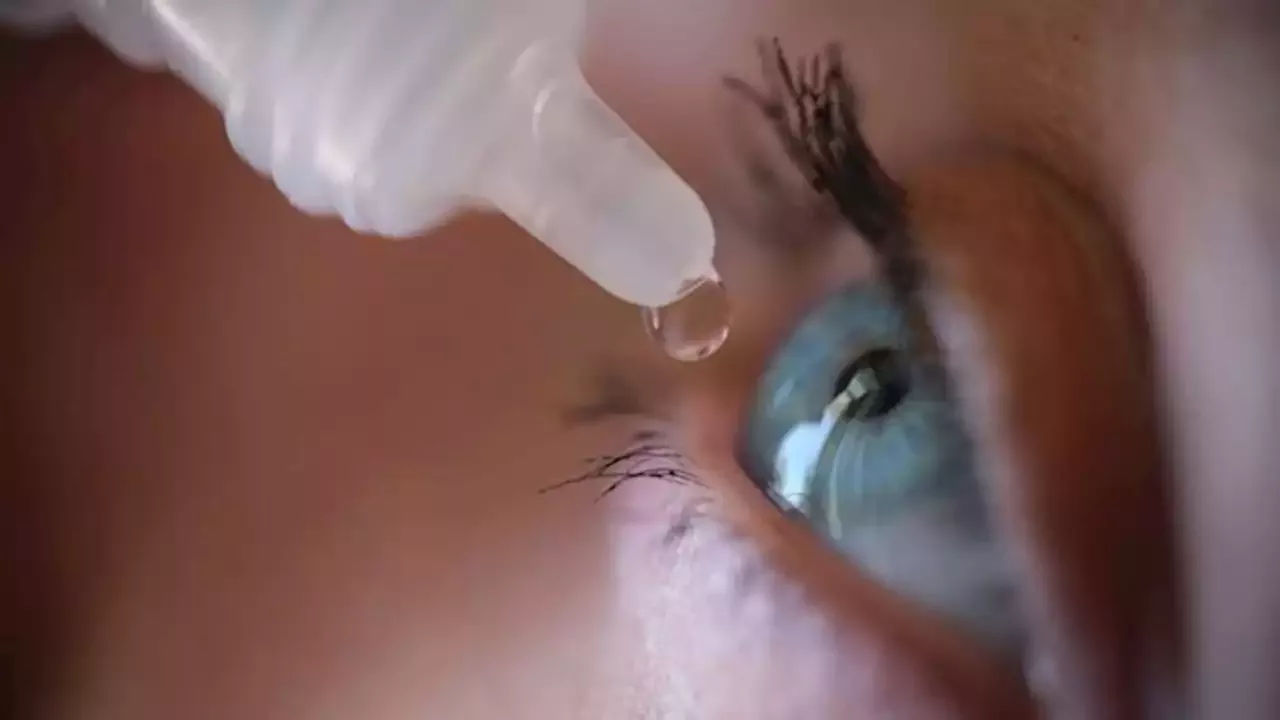
In their letters, the FDA also said people using these unapproved eye products that claim to treat or cure certain conditions may cause delay or stoppage of treatments that are approved by the agency. The FDA told the companies they have 15 days to respond to the FDA in writing to address any steps they have taken to correct violations.
대한민국 최근 뉴스, 대한민국 헤드 라인
Similar News:다른 뉴스 소스에서 수집한 이와 유사한 뉴스 기사를 읽을 수도 있습니다.
 FDA sends letters to CVS, Walgreens, other companies about unapproved eye productsThe FDA sent warning letters to eight companies, including CVS and Walgreens, warning them against making or selling unapproved eye products.
FDA sends letters to CVS, Walgreens, other companies about unapproved eye productsThe FDA sent warning letters to eight companies, including CVS and Walgreens, warning them against making or selling unapproved eye products.
더 많은 것을 읽으십시오 »
 FDA issues warning letters to CVS, Walgreens, other companies over unapproved eye productsThe FDA sent warning letters to eight companies, including CVS and Walgreens, warning them against making or selling unapproved eye products.
FDA issues warning letters to CVS, Walgreens, other companies over unapproved eye productsThe FDA sent warning letters to eight companies, including CVS and Walgreens, warning them against making or selling unapproved eye products.
더 많은 것을 읽으십시오 »
 FDA sends letters to CVS, Walgreens, other companies about unapproved eye productsThe FDA sent warning letters to eight companies, including CVS and Walgreens, warning them against making or selling unapproved eye products.
FDA sends letters to CVS, Walgreens, other companies about unapproved eye productsThe FDA sent warning letters to eight companies, including CVS and Walgreens, warning them against making or selling unapproved eye products.
더 많은 것을 읽으십시오 »